Transition metals reactivity2/29/2024  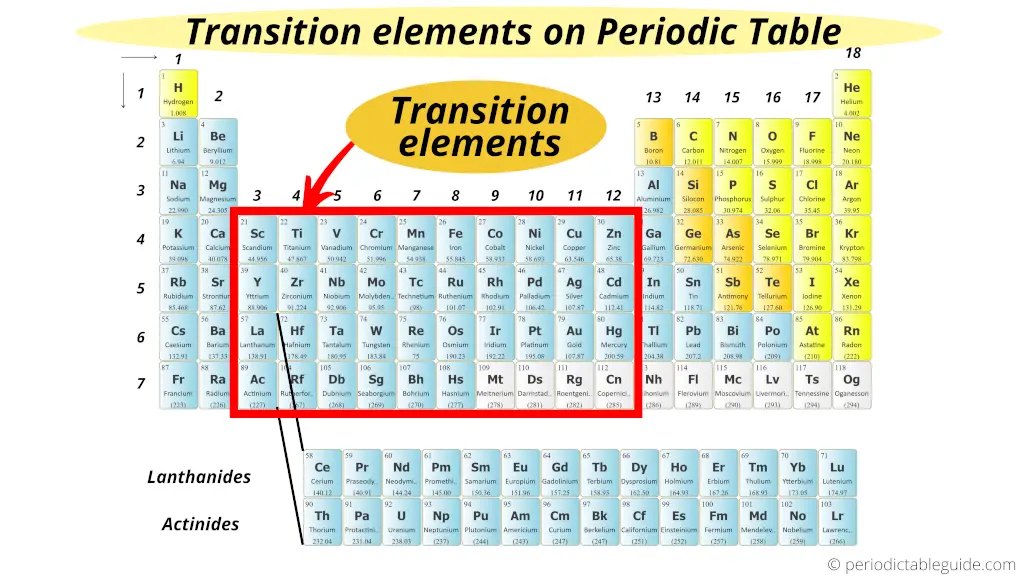 
Looking for something specific in this course? The Resource Index compiles links to most course resources in a single page. Lecture 29: Transition Metals: Crystal Field Theory Part II Lecture 28: Transition Metals: Crystal Field Theory Part I Lecture 27: Introduction to Transition Metals Lecture 26: Chemical and Biological Oxidations Lecture 25: Oxidation-Reduction and Electrochemical Cells By the end of the unit, viewers should be able to calculate electrochemical cell potentials, identity which compound is the better oxidizing agent, draw energy diagrams that explain why one cobalt compound is blue and another red, and explain how a chelator works to treat acute lead poisoning. Viewers can observe some of these special properties for themselves as spectacular color changes afforded by solutions of nickel and cobalt salts are demonstrated. Revisiting the concept of atomic orbitals, we discuss how the occupation of electrons in d-orbitals gives rise to the special properties of d-block metal-containing complexes. Whereas metals in group I and II of the periodic table are only oxidized by one and two electrons, respectively, transition (d-block) metals are capable of achieving multiple oxidation states. For example, the most stable compounds of chromium are those of Cr(III), but the corresponding Mo(III) and W(III) compounds are highly reactive. In Unit IV, we continue to consider chemical reactions but now focus on reactions that involve the loss (oxidation) and gain (reduction) of electrons. For example, the HgC bond of (MeHg) + cation is indefinitely stable in aqueous H 2 SO 4 solution in air. On the contrary the late transition metal organometallic compounds are much less reactive and stable. This Special Issue is devoted to all aspects of transition metal chemistry. It is closely related to other fields such as supramolecular chemistry, photochemistry, catalysis, medicine, materials science, quantum chemistry, etc. Despite their long history in catalysis, the discovery of new. Some of the early transition metal catalyzed reactions are still being used today. Finally, we extend our analysis to ternary transition-metal alloys and identify the most resilient compounds to O and S binding.Unit IV: Transition Metals & Oxidation-Reduction Reactions Similar high reactivity was also observed in case of the early transition metal organometallic compounds particularly of Ti and Zr. The coordination chemistry of transition metals is one of the most important and extensive areas of modern chemistry. Transition metals make excellent catalysts as they have incompletely filled d-orbitals that allow them to both donate and accept electrons easily from other molecules. Such descriptor is then applied to a vast database of electronic structures of transition-metal alloys, for which we are able to predict the range of binding energies across both the compositional and the structural space. This is here obtained by fitting several schemes, based on the original Newns-Anderson model, against density-functional-theory data for the 4$d$ transition metal series. The analysis is performed by defining a suitable descriptor for the binding energy. With this in mind, we present a systematic study of the binding energy of O and S across the entire transition-metals composition space, namely we explore the binding energy of transition-metal binary alloys. These are always initiated with O and S binding to a metallic surface, so that one can use the binding energy as a rough proxy for the metal reactivity. Download a PDF of the paper titled Reactivity of transition-metal alloys to oxygen and sulphur, by Rajarshi Tiwari and 2 other authors Download PDF Abstract:Oxidation and tarnishing are the two most common initial steps in the corrosive process of metals at ambient conditions.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
